
Octopus Distribution provides GDP- and GDPMD-compliant cold chain logistics in Malaysia tailored for the stringent demands of the pharmaceutical sector. Our end-to-end cold chain capabilities integrate validated temperature-controlled warehousing, qualified cold box, and regulatory-aligned handling protocols to safeguard product stability throughout the supply chain.
With advanced cold chain management for pharmaceuticals, every shipment is monitored in real time using our own proprietary warehouse management system (WMS). Whether supporting vaccine distribution, high-value biologics, or time and temperature sensitive products, we deliver complete product traceability and quality assurance from origin to point-of-use.
Why Is Cold Chain Important to the Pharmaceutical Industry
Cold chain logistics in Malaysia ensures temperature-sensitive products remain safe, effective, and compliant from origin to final delivery. At Octopus Distribution, our certified operations work by protecting the integrity of medicines, active ingredients, and medical devices by maintaining strict temperature controls from warehousing to transport.
With GDP-, WHO-, and IATA-aligned cold chain management for pharmaceuticals, we combine advanced temperature monitoring, calibrated storage facilities, and validated transport solutions to eliminate the risk of costly product loss or regulatory breaches.
With GDP-, WHO-, and IATA-aligned cold chain management for pharmaceuticals, we combine advanced temperature monitoring, calibrated storage facilities, and validated transport solutions to eliminate the risk of costly product loss or regulatory breaches.
How We Deliver Cold Chain Integrity for Pharmaceuticals
Our cold chain management framework maintains product integrity across storage, handling, and transportation. Each stage is validated, documented, and monitored in real time to meet compliance requirements and customer specifications.
Our Temperature Profiles & Storage Capabilities
Octopus Distribution maintains validated temperature zones for pharmaceutical and medical device storage, supported by continuous monitoring and calibration to meet GDP and GDPMD standards. Each temperature profile is designed to protect product stability and comply with manufacturer specifications.

CRT: 18°C to 25°C
Cold Room Temperature (CRT) is ideal for most pharmaceuticals, including oral medications and general medical supplies. Warehouses are equipped with temperature sensors and data loggers to maintain stable conditions.

Cold Storage: 2°C to 8°C
Used for vaccines, insulin, and other biologics requiring chilled storage. Backup power systems and redundancy protocols prevent temperature fluctuations.
Who We Serve

Pharmaceutical Manufacturers
Secure storage and delivery of temperature-sensitive finished products.

Veterinarians and Animal Health Providers
Reliable cold chain handling for veterinary medicines, vaccines, and other temperature-sensitive animal healthcare products.
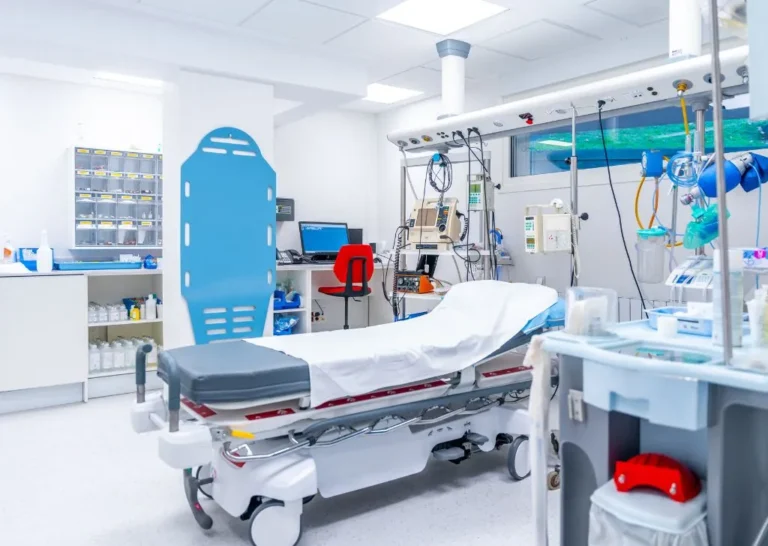
Hospitals and Healthcare Facilities
Reliable supply of essential medicines and cold chain medical devices.

Clinical Research Organisations (CROs)
Controlled storage and transport for investigational drugs and trial materials.
Medical Device Companies
Compliance-ready handling for temperature-sensitive devices and diagnostic kits.

Government Health Agencies
Large-scale public health and community healthcare distribution programmes.
By aligning operational execution with cold chain management for pharmaceuticals, we help our clients protect product integrity, maintain compliance, and deliver with confidence.
Why Should You Choose Octopus Distribution As Your Preferred Cold Chain Logistics Partner in Malaysia?
In a highly regulated and competitive landscape, what sets leading cold chain logistics in Malaysia apart is our ability to deliver consistent compliance, secure handling, and scalable logistics infrastructure. At Octopus Distribution, we offer solutions that seamlessly integrate into your supply chain as a trusted regulatory and logistics partner.
Certified Warehousing & GDP/ISO/GDPMD Compliance
Cold Chain & Poison Storage Expertise
End-to-end Logistics, From Import to Final-Mile Delivery
Frequently Asked Questions (FAQ)
What pharmaceutical products require cold chain logistics?
Most vaccines, biologics, insulin, blood products, and certain active pharmaceutical ingredients require cold chain handling. Some medical devices with temperature-sensitive components also fall into this category.
How does Octopus ensure temperature integrity during transport?
We use calibrated temperature-controlled vehicles, validated packaging, and real-time monitoring. Any deviation triggers immediate corrective action in line with GDP and GDPMD protocols.
Can Octopus handle cold chain deliveries to East Malaysia?
Yes. We operate nationwide delivery, including to Sabah and Sarawak, with temperature-controlled shipping and validated distribution routes.
How do you ensure consistent temperature in every shipment?
Every cold chain shipment is equipped with a WarmMark temperature sensor to ensure your goods stay within our 18°C to 25°C and 2°C to 8°C range during loading and transportation.
How do you manage temperature excursions or delays?
We have documented contingency procedures, including backup storage, route diversion, and product isolation protocols to maintain product quality and compliance.
Can Octopus Distribution do cold chain logistics for food products?
No. Octopus Distribution focuses exclusively on pharmaceuticals, medical devices, and healthcare-related products. We do not handle cold chain logistics for food, beverages, or perishable consumer goods.
Partner with Octopus Distribution for Reliable Cold Chain Logistics
When your pharmaceuticals or medical devices require strict temperature control, Octopus Distribution delivers the infrastructure, compliance, and expertise to protect your products from start to finish.
We operate certified cold chain facilities, qualified cold box, and real-time monitoring systems to ensure every shipment meets GDP and GDPMD requirements.
Contact us today to discuss your pharmaceutical cold chain requirements or request a tailored proposal. Our team will assess your needs and provide a solution that meets both regulatory standards and operational deadlines.
















