
Transport is the most vulnerable stage of the cold chain. In a warehouse, temperature conditions are controlled, monitored, and backed up by redundant systems. On the road, the variables multiply and the window for intervention narrows considerably.
For pharmaceutical and medical device companies operating in Malaysia, where ambient temperatures regularly exceed 33°C and urban congestion is a daily operational reality, cold chain transport is a critical quality event that occurs every time a temperature-sensitive product leaves a facility and travels to a hospital, clinic, or end user.
This article covers what robust cold chain transport management looks like in practice, where the most common failure points occur, and what the consequences are when the cold chain breaks for your product, your regulatory standing, and ultimately for the patients your products serve.
What “Maintaining the Cold Chain” Actually Means in Transport
Maintaining the cold chain during transport means ensuring that a temperature-sensitive product remains within its required temperature range (continuously, without interruption) from the moment it leaves a controlled storage environment to the moment it is received and stored at its destination.
For most pharmaceutical products, that range is 2–8°C. For frozen biologics and certain vaccines, it may be -20°C or lower. For some medical devices and reagents, it may be a controlled ambient range of 15–25°C. In every case, the requirement is the validated condition under which the product’s safety, efficacy, and regulatory status are guaranteed.
Maintaining that condition during transport requires active management across four distinct stages: pre-transport preparation, active transit, transition points, and receipt at destination. Each stage carries its own failure modes, and a breakdown at any one of them constitutes a cold chain excursion regardless of how well the others were managed.
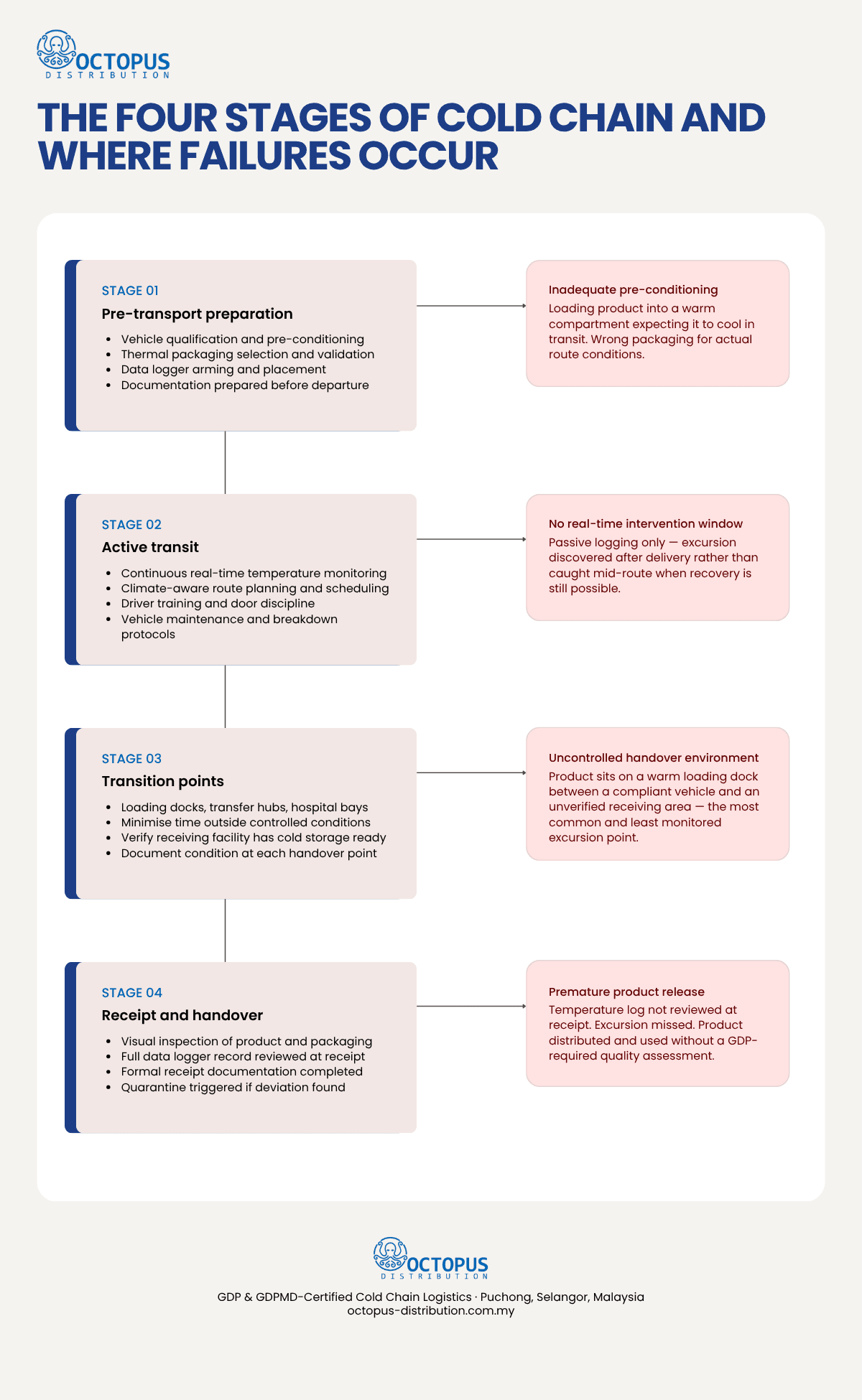
Stage One: Pre-Transport Preparation
The cold chain during transport is largely determined before the vehicle moves. Preparation failures are the most preventable category of cold chain breakdown, and also among the most common.
Vehicle qualification and pre-conditioning
A refrigerated vehicle must be qualified for the temperature range it is carrying — not just equipped with a cooling unit, but validated to demonstrate it can maintain the required range under real operating conditions.
Before loading, the vehicle’s cold compartment should be pre-conditioned to the target temperature range. Loading a product into a warm compartment and expecting it to cool down in transit is a significant and common error, particularly in warm-weather markets like Malaysia where ambient temperatures give a warm vehicle a substantial thermal head start.
Packaging selection and validation
Thermal packaging such as insulated shippers, gel packs, dry ice, and phase change materials must be validated for the specific temperature range, the expected transit duration, and the anticipated ambient conditions. A validated shipper rated for a 24-hour transit at 25°C ambient performs differently in a 35°C Malaysian afternoon. Packaging validation data should match the real-world conditions of the route, not laboratory conditions that may not reflect the operational environment.
Documentation before departure
GDP and GDPMD guidelines require that products in transit are accompanied by accurate documentation including product identity, quantity, storage conditions, and any relevant handling instructions.
Temperature data loggers should be armed and placed correctly before loading, not after. A logger that starts recording 20 minutes into the journey has already missed the loading event, which is frequently the moment when the first excursion occurs.
Stage Two: Active Transit
Once a product is in transit, the primary tools for maintaining the cold chain are vehicle performance, routing discipline, and real-time monitoring.
Continuous temperature monitoring
Modern cold chain transport relies on electronic temperature data loggers or telematics-integrated sensors that record temperature at defined intervals, typically every 5 to 15 minutes, throughout the journey.
For high-value or high-risk products, real-time transmission of temperature data to a central monitoring system allows alerts to be triggered and response protocols activated while the vehicle is still on the road. This is the difference between catching an excursion in progress and discovering one after delivery.
Route planning with thermal risk in mind
Not all routes carry equal cold chain risk. Urban routes with frequent stops, extended idling in traffic, and multiple door-opening events accumulate thermal load over the course of a delivery day. Routes that pass through areas with limited service infrastructure where a vehicle breakdown would leave a product stranded for hours in the heat carry a different risk profile than controlled point-to-point runs.
Route planning accounts for these variables, scheduling the most temperature-sensitive deliveries during cooler morning windows and avoiding high-risk routes where alternatives exist.
Driver training and operational discipline
The behaviour of the driver at each delivery point has a direct impact on temperature maintenance.
- Every door opening introduces ambient air into the cold compartment.
- Extended stops with the engine off can reduce or eliminate active cooling.
- Improper stacking or overloading can restrict airflow around products and create warm pockets within an otherwise controlled load.
These are routine operational variables that determine whether a compliant shipment remains compliant across a full delivery run.
Vehicle maintenance and contingency protocols
A refrigeration unit that fails mid-route is a foreseeable risk, not an unforeseeable event. Preventive maintenance schedules for refrigeration units, documented contingency procedures for breakdowns, and pre-arranged backup vehicle availability are elements of a mature cold chain transport operation. In Malaysia’s heat, a refrigeration failure without a rapid response protocol is not a recoverable situation for most pharmaceutical products.
Stage Three: Transition Points
Transition points like loading docks, intermediate transfer facilities, and hospital receiving bays are where cold chain failures are most likely to occur and least likely to be monitored.
For example, a product that maintains 4°C throughout a two-hour vehicle journey can experience a significant excursion in the ten minutes it sits on an uncontrolled loading dock waiting for a receiving clerk.
Transition risk management involves minimising the time a product spends outside controlled conditions at each handover point, ensuring that receiving facilities have appropriate cold storage available before delivery is attempted, and documenting the condition of the product, including a temperature log review, at each transfer point. Where receiving conditions are substandard or unknown, validated passive packaging must be capable of maintaining the required temperature range for the full expected exposure period, with margin.
In practice, this means that a cold chain logistics provider must have visibility and protocols not just for the journey itself, but for what happens at both ends of it. Delivering a product in perfect condition to a hospital receiving area that lacks refrigerated intake storage is not a successful cold chain delivery.
Stage Four: Receipt and Handover
The cold chain is not complete until the product has been received into a qualified storage environment and the temperature record for the full journey has been reviewed and accepted. Receipt procedures should include a visual inspection of the product and packaging, review of the data logger record for any excursions during transit, and formal documentation of the receipt including any deviations noted and the risk assessment applied.
Where a temperature deviation is identified at receipt, the product should be quarantined pending investigation. GDP guidelines are explicit that products which have experienced unvalidated temperature excursions must not be released for use without a formal quality assessment.
What Happens If the Cold Chain Breaks
A cold chain excursion triggers a cascade of consequences whose severity depends on the product involved, the magnitude and duration of the deviation, and the robustness of the documentation and response.
Product degradation and loss of efficacy
The most direct consequence of a cold chain break is damage to the product itself. Vaccines exposed to temperatures outside 2–8°C can lose potency irreversibly. There is no way to restore efficacy once thermal damage has occurred.
Biologics, insulin, and monoclonal antibodies are similarly sensitive. The damage is not always visible: a product that has been temperature-compromised may look, smell, and handle identically to an intact product. Without a complete temperature record, there is no way to distinguish between the two.
Patient safety risk
When a compromised product reaches a patient — a vaccine that no longer provides immunity, a biologic that has partially degraded, a diagnostic reagent that produces inaccurate results — the consequences extend far beyond the commercial transaction.
The patient who receives an ineffective vaccine because a cold chain broke somewhere between the manufacturer and the clinic has no visibility into what went wrong. The responsibility sits entirely with the supply chain.
Regulatory investigation and compliance consequences. Under Malaysia’s GDP and GDPMD frameworks, a temperature excursion during transport is a quality event that must be documented, investigated, and risk-assessed. If the product was released without a proper assessment, or if the documentation trail is incomplete, the compliance consequences can extend well beyond the immediate event to facility audits, licence reviews, and in serious cases, product recalls.
Product recall
Where an excursion is identified after a product has been distributed and released for use, a recall may be required. The scope of a recall is determined by the traceability of the affected batch including how many units were distributed, to how many locations, and over what time period. A distribution partner with robust batch tracking and serialisation can contain a recall to the affected units. One without it faces a much broader and more costly recall scope.
Financial exposure
The commercial cost of a cold chain failure compounds quickly. Product loss, investigation costs, re-supply expenses, recall logistics, regulatory engagement, and the downstream damage to customer relationships with hospitals and clinics each add a layer to the total exposure. As discussed in our article on climate change and cold chain risk, the total cost of a single excursion event is typically five to ten times the value of the product itself once all downstream consequences are included.
Reputational damage
For pharmaceutical and medical device companies, reputation is built on the reliability and safety of products in the hands of healthcare providers. A single publicised cold chain failure can damage relationships with hospital procurement teams and medical practitioners that took years to establish. In a market like Malaysia, where referral networks and professional trust are central to commercial relationships in healthcare, reputational damage is a long-term business risk, not just a short-term commercial setback.
The Standard Every Cold Chain Transport Partner Should Meet
Cold chain transport management is ultimately a function of two things: the systems and infrastructure a logistics partner operates, and the discipline with which their teams execute at every stage of the journey. Neither is sufficient without the other.
A GDP and GDPMD-certified distribution partner brings validated vehicles, qualified packaging, real-time monitoring, and documented response protocols but the value of those capabilities is only realised if they are applied consistently, across every shipment, on every route, by every driver and warehouse operative involved in the cold chain.
At Octopus Distribution, cold chain transport is managed as a quality function from pre-shipment preparation through to confirmed receipt. Our facilities operate under GDP, GDPMD, and ISO 9001 certification, and our customised warehouse management system provides end-to-end temperature visibility with automated alerting and a complete, tamper-evident audit trail for every product movement.
Cold chain integrity during transport is not a complex problem but it is an unforgiving one. The cost of getting it wrong, for the product, the patient, and the business, is always higher than the cost of getting it right.
Octopus Distribution provides GDP and GDPMD-certified cold chain logistics across Malaysia, serving pharmaceutical and medical device companies who require end-to-end temperature assurance from storage through to last-mile delivery. Contact our team to discuss your cold chain transport requirements.










